Abstract
Diffuse midline gliomas (DMGs) harboring the H3K27M mutation represent a rare and aggressive category of central nervous system tumors recently classified as WHO grade IV. While predominantly observed in pediatric brainstem gliomas, spinal cord involvement, especially in adults, is uncommon and presents unique diagnostic and therapeutic challenges. We report a case involving a 27-year-old male patient who presented with progressively worsening back pain and sensory disturbances localized below the T12 dermatome. Magnetic resonance imaging (MRI) revealed an expansive intramedullary lesion in the thoracic spine (T10–T12). Surgical intervention achieved a gross total resection, and histopathological evaluation confirmed a DMG characterized by strong positivity for the H3K27M mutation, an elevated Ki-67 proliferation index, and significant p53 expression. Postoperatively, the patient underwent adjuvant radiotherapy (54 Gy) and initiated temozolomide chemotherapy. A follow-up MRI at five months post-treatment showed no evidence of recurrence. Spinal cord DMGs with the H3K27M mutation typically exhibit aggressive biological behavior and predominantly affect individuals under the age of 40. These tumors commonly exhibit high proliferative activity and lack MGMT promoter methylation, suggesting a limited effectiveness of conventional alkylating agents, such as temozolomide. Although gross total resection alone is rarely curative, integrated multimodal therapy can provide meaningful short-term disease control. Continued research into targeted epigenetic therapies is crucial for advancing management and improving patient outcomes.
Keywords: diffuse midline glioma, H3K27M mutation, spinal cord tumor, intramedullary glioma, WHO grade 4 glioma
Introduction
Diffuse midline glioma (DMG), H3K27-altered, is classified as a World Health Organization (WHO) Grade IV tumor entity, characterized by a critical lysine-to-methionine substitution at position 27 on histone H3 (H3K27M). Initially identified predominantly in pediatric brainstem gliomas, these tumors have been increasingly recognized in other midline central nervous system (CNS) structures, including the thalamus and spinal cord. Their aggressive biological behavior contributes significantly to poor prognosis, and the complex genetic and epigenetic underpinnings pose considerable therapeutic challenges.1,2
While spinal cord presentations of DMG remain exceptionally rare, particularly in adults, accurate diagnosis requires heightened clinical suspicion complemented by advanced imaging, histopathological evaluation, and precise molecular characterization. The presence of the H3K27M mutation, which disrupts normal epigenetic regulation by inhibiting the Polycomb Repressive Complex 2 (PRC2), plays a pivotal role in tumor progression and resistance to conventional therapies. Understanding the implications of these molecular characteristics is crucial, as emerging targeted therapeutic strategies increasingly aim to modulate these specific epigenetic abnormalities.3-5
This case report presents a rare instance of spinal H3K27M-mutant glioma in an adult patient, providing insights into its clinical course, diagnostic intricacies, and therapeutic management. Furthermore, a comprehensive review of the existing literature is provided to contextualize clinical presentations, molecular diagnostic criteria, current treatment paradigms, and potential future directions for targeted therapeutic interventions.
Case Presentation
A 27-year-old male presented with a one-week history of progressive upper and lower back pain, following an initial two-week period of mild discomfort. The neurological examination revealed hypoesthesia localized to the T12 dermatome and below, without associated motor deficits or other neurological abnormalities. Magnetic resonance imaging (MRI) demonstrated an intramedullary expansile lesion extending from T10 to T12, characterized by T2-weighted hyperintensity, cord swelling, and surrounding edema. Post-contrast T1 sequences displayed mild, patchy enhancement suggestive of a high-grade glial neoplasm (Figure 1).
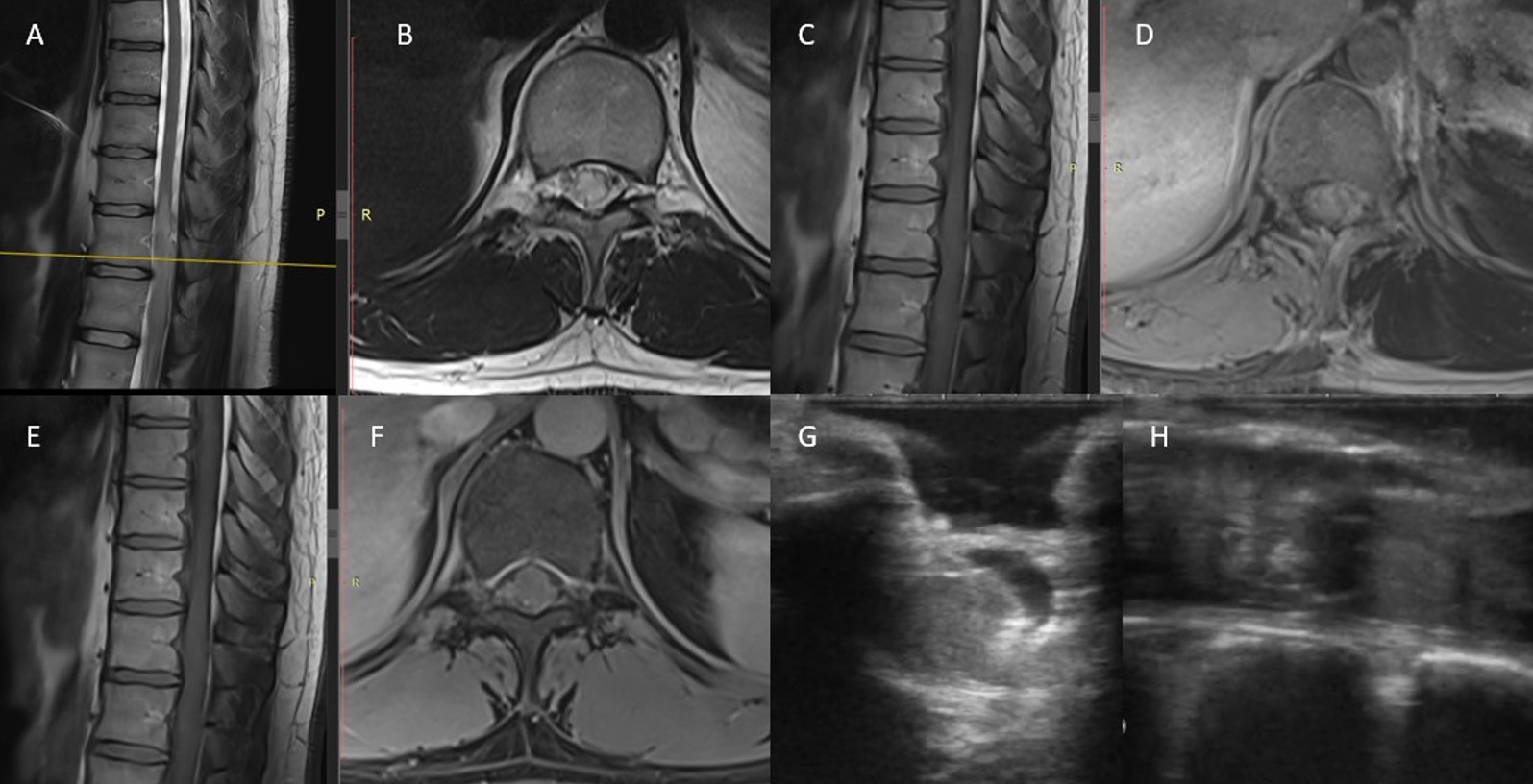
The patient underwent surgical resection under general anesthesia in the prone position. Using fluoroscopic guidance, the T10–12 vertebral levels were localized, and a midline incision was performed. Paraspinal muscles were dissected bilaterally, and a laminectomy was carried out to expose the dura. Upon dural opening, a mass consistent with tumor tissue was identified intradurally. Gross total resection of the lesion was achieved using bipolar cautery, tumor forceps, and Cavitron Ultrasonic Surgical Aspirator (CUSA). Dural closure was completed primarily, and meticulous hemostasis was secured using a Duragen patch and fibrin sealant. Layered anatomical closure of the surgical wound followed (Figure 2).
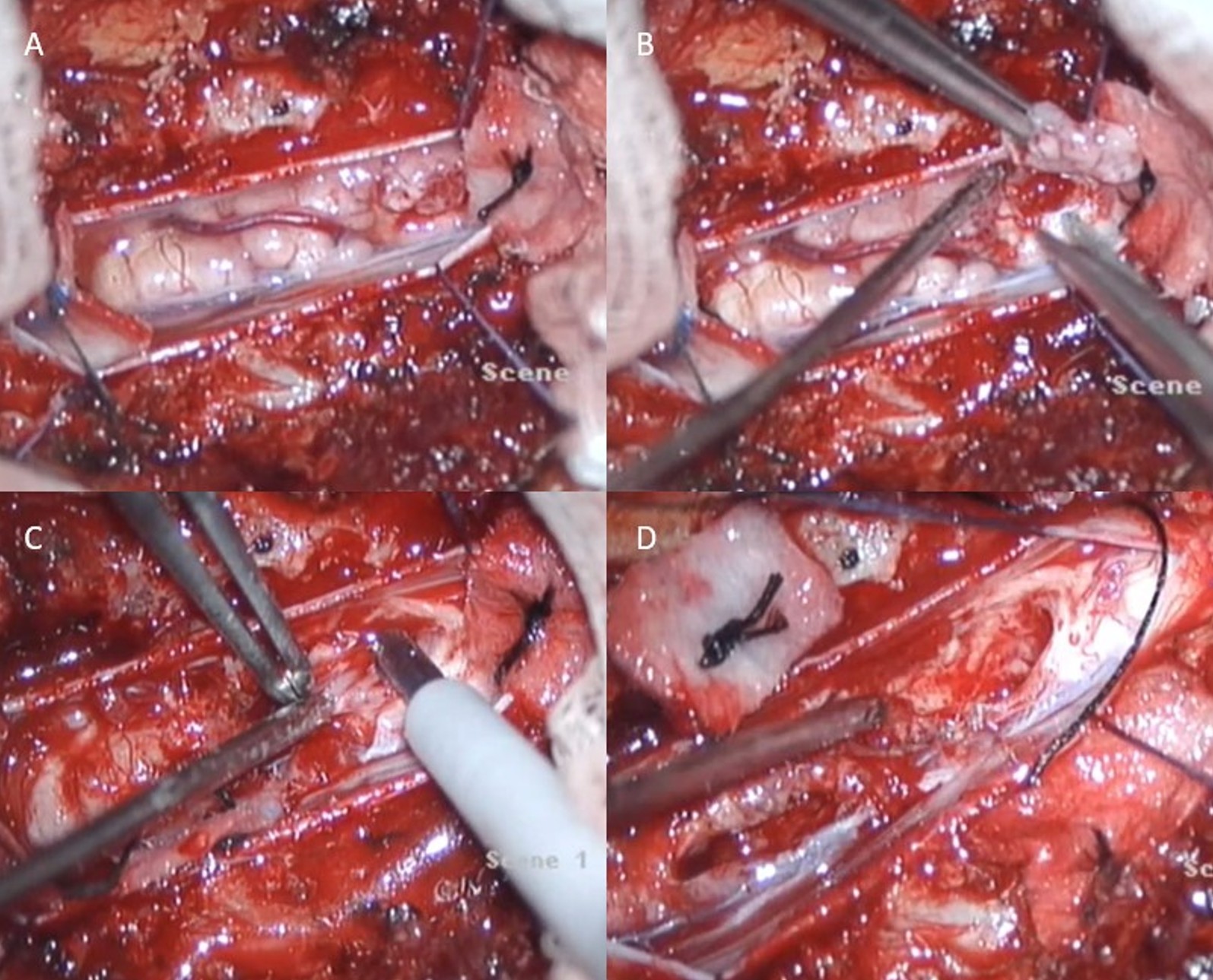
Postoperative neurological status remained stable, with persistent hypoesthesia but no additional deficits. The patient subsequently received adjuvant radiotherapy totaling 54 Gy and was placed on maintenance temozolomide therapy. Follow-up MRI performed five months postoperatively showed no evidence of tumor recurrence (Figure 3).
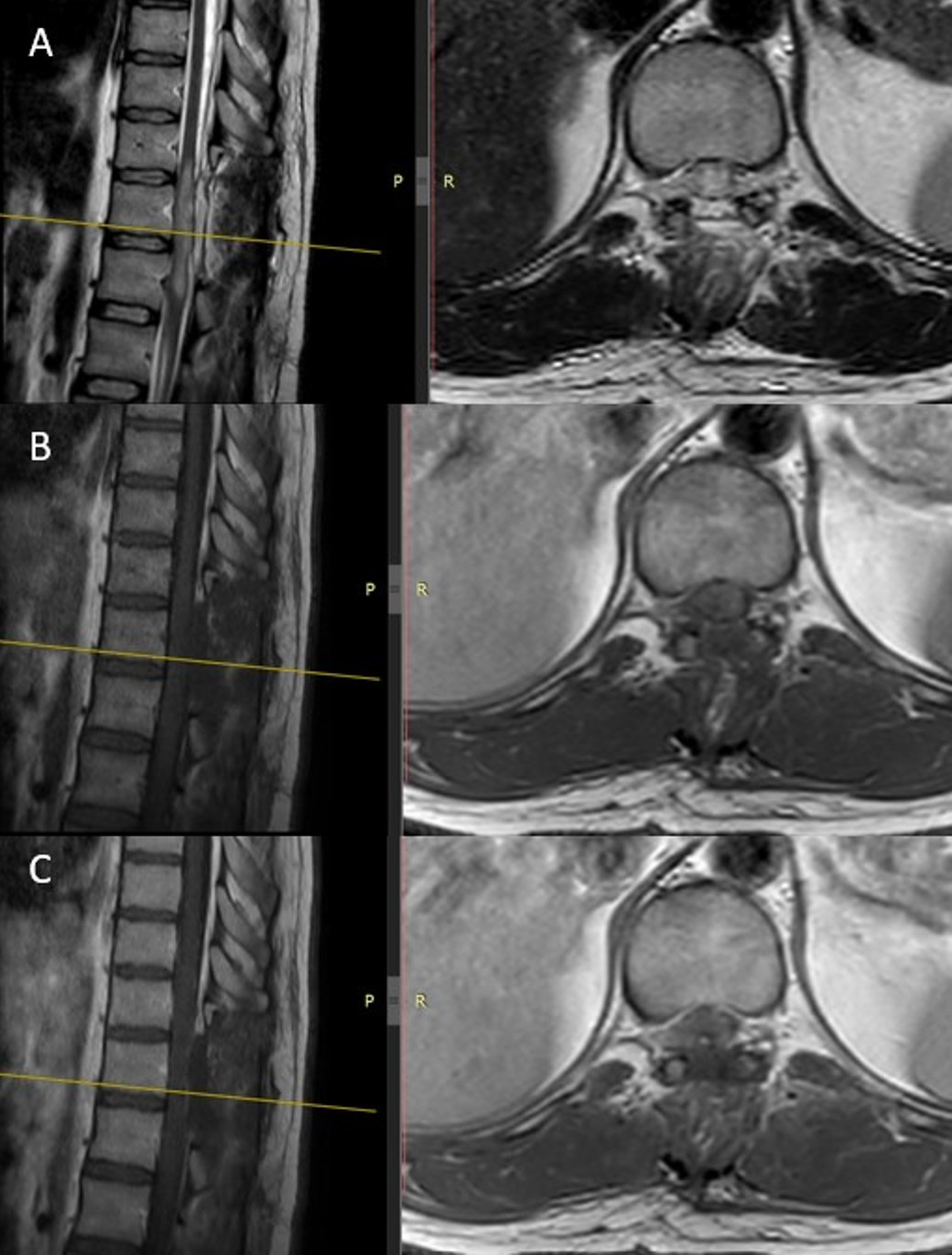
Histopathology and molecular profile
Histopathological examination of the resected spinal mass revealed a diffusely infiltrative glial tumor. Microscopically, the neoplasm exhibited moderate nuclear atypia, hyperchromasia, and scattered mitotic activity. Areas of necrosis and focal microvascular proliferation were also observed. Immunohistochemical staining was diffusely positive for glial fibrillary acidic protein (GFAP), confirming its astrocytic origin. Importantly, strong positivity for the H3K27M mutant protein was detected, establishing the diagnosis of diffuse midline glioma, H3K27-altered, WHO grade IV.
The Ki-67 proliferation index was elevated at approximately 15%, indicating a high proliferative potential. Additional molecular analyses showed wild-type status for IDH1/2, retained ATRX expression, and absence of the BRAF V600E mutation. Increased p53 immunoreactivity suggested a TP53 mutation. The MGMT promoter was found to be unmethylated, further indicating likely resistance to alkylating chemotherapy. Testing for TERT promoter mutations was not performed.
These molecular characteristics align closely with established profiles of spinal diffuse midline gliomas harboring the H3K27M mutation, which typically exhibit aggressive clinical behavior and limited responsiveness to standard therapeutic approaches.
Discussion
Diffuse midline gliomas (DMGs) with H3K27M-alteration represent a rare and aggressive category of central nervous system (CNS) neoplasms predominantly observed in pediatric brainstem locations. Spinal cord involvement, particularly in adults, remains an uncommon and distinct presentation, necessitating careful differentiation from other spinal intramedullary tumors. This case of a 27-year-old male with a thoracic intramedullary DMG underscores critical aspects of clinical presentation, diagnostic challenges, and management paradigms of this complex entity.3,6,7
Radiologically, DMGs share imaging characteristics with other spinal cord tumors such as astrocytomas, ependymomas, hemangioblastomas, and demyelinating diseases. Typical MRI findings include diffuse cord expansion, hyperintense signaling on T2-weighted images, and variable enhancement patterns.8 The presence of significant peritumoral edema, as observed in our case, often raises suspicion for high-grade gliomas. Consistent with this, Watanabe et al. highlighted thoracic involvement as a predominant spinal localization, supporting the representativeness of our patient’s presentation.3
Molecular profiling has become indispensable in diagnosing and prognosticating DMGs. The hallmark mutation, H3K27M, results in profound epigenetic dysregulation and significantly contributes to tumor aggressiveness. Our patient’s tumor displayed classic molecular features, including H3K27M positivity, wild-type IDH status, high Ki-67 proliferation rate, and lack of MGMT promoter methylation, corroborating previous literature. The absence of MGMT methylation, in particular, implies resistance to alkylating chemotherapeutic agents, which explains the limited clinical benefit from temozolomide.9,10
Surgical management strategies remain debated, particularly regarding the extent of resection. While gross total resection is typically associated with better outcomes in other CNS tumors, evidence suggests it may not significantly impact survival in spinal DMGs due to inherent microscopic infiltrative growth patterns. In our case, gross total resection was achievable due to clear tumor margins and lesion accessibility. However, contemporary studies suggest equivalent or superior outcomes with limited surgical intervention combined with adjuvant radiotherapy and chemotherapy, underscoring a conservative surgical philosophy.11,12
The role of chemotherapy, primarily temozolomide, remains uncertain due to the intrinsic resistance mechanisms.13,14 Novel agents targeting specific molecular pathways disrupted by the H3K27M mutation, such as histone deacetylase inhibitors (panobinostat), dopamine receptor antagonists (ONC201), and anti-angiogenic therapies (bevacizumab), have shown preliminary promise in preclinical and early clinical trials. Further rigorous evaluation through well-designed prospective trials is critical.15
Our patient’s favorable short-term outcome highlights the potential efficacy of a comprehensive multimodal therapeutic strategy, including surgery, radiotherapy, and chemotherapy. Nonetheless, the prognosis remains guarded given the aggressive biological behavior of spinal DMGs.
Accordingly, longer-term follow-up and larger, systematically analyzed case series are required to clarify the true prognostic value of surgical extent and to better define optimal management strategies for spinal H3K27-altered diffuse midline gliomas. In parallel, future research should prioritize large-scale, multicenter studies incorporating standardized molecular profiling, centralized clinical registries, and the systematic evaluation of targeted therapies aimed at modulating the epigenetic dysregulation central to these tumors, with the potential to substantially improve disease understanding, patient management, survival, and quality of life.
Conclusion
Diffuse midline gliomas (DMGs) with H3K27M mutation in the spinal cord are rare and aggressive tumors that predominantly affect young adults, presenting significant diagnostic and therapeutic challenges. Although universally categorized as WHO grade IV, emerging evidence suggests potential variability in clinical outcomes. Comprehensive molecular profiling is crucial for accurate diagnosis and informing targeted treatment strategies. While the role of aggressive surgical resection remains controversial, a multimodal therapeutic approach incorporating surgery, radiotherapy, and chemotherapy may offer short-term disease control. Future research should focus on multicenter collaboration, detailed molecular stratification, and the development and evaluation of novel targeted therapies aimed at mitigating the aggressive biology associated with H3K27M mutations, ultimately aiming to enhance patient outcomes and survival.
Acknowledgment
The authors would like to thank the Department of Pathology and the Department of Radiology for their contributions to the diagnosis and documentation of this case. We also acknowledge the patient and his family for providing consent to share clinical data for educational and research purposes.
Ethical approval
This study is a case report, therefore does not require an ethics committee approval.
Source of funding
The authors declare the study received no funding.
Conflict of interest
The authors declare that there is no conflict of interest.
References
- Louis DN, Perry A, Wesseling P, et al. The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol 2021; 23: 1231-1251. https://doi.org/10.1093/neuonc/noab106
- Louis DN, Giannini C, Capper D, et al. cIMPACT-NOW update 2: diagnostic clarifications for diffuse midline glioma, H3 K27M-mutant and diffuse astrocytoma/anaplastic astrocytoma, IDH-mutant. Acta Neuropathol 2018; 135: 639-642. https://doi.org/10.1007/s00401-018-1826-y
- Watanabe G, Wong JM, Estes B, et al. Diffuse midline H3K27-altered gliomas in the spinal cord: a systematic review. J Neurooncol 2024; 166: 379-394. https://doi.org/10.1007/s11060-024-04584-8
- Luo Y, Zeng L, Xie XQ, et al. H3K27M mutant diffuse midline glioma: a case report. Eur Rev Med Pharmacol Sci 2020; 24: 2579-2584. https://doi.org/10.26355/eurrev_202003_20527
- Bhattarai AM, Mainali G, Jha P, et al. Diffuse midline glioma H3K27M mutation in adult: a case report. Ann Med Surg (Lond) 2022; 76: 103567. https://doi.org/10.1016/j.amsu.2022.103567
- Yekula A, Gupta M, Coley N, U HS. Adult H3K27M-mutant diffuse midline glioma with gliomatosis cerebri growth pattern: Case report and review of the literature. Int J Surg Case Rep 2020; 68: 124-128. https://doi.org/10.1016/j.ijscr.2020.02.046
- Navarro RE, Golub D, Hill T, et al. Pediatric midline H3K27M-mutant tumor with disseminated leptomeningeal disease and glioneuronal features: case report and literature review. Childs Nerv Syst 2021; 37: 2347-2356. https://doi.org/10.1007/s00381-020-04892-0
- Houten JK, Weiner HL. Pediatric intramedullary spinal cord tumors: special considerations. J Neurooncol 2000; 47: 225-230. https://doi.org/10.1023/a
- Handis C, Tanrıkulu B, Danyeli AE, Özek MM. Spinal intramedullary H3K27M mutant glioma with vertebral metastasis: a case report. Childs Nerv Syst 2021; 37: 3933-3937. https://doi.org/10.1007/s00381-021-05119-6
- Aftahy AK, Butenschoen VM, Hoenikl L, et al. A rare case of H3K27-altered diffuse midline glioma with multiple osseous and spinal metastases at the time of diagnosis. BMC Neurol 2023; 23: 87. https://doi.org/10.1186/s12883-023-03135-4
- Griessmair M, Delbridge C, Zimmer C, et al. Case report: an unusual long-term evolution of a diffuse midline glioma, H3K27 altered. Front Oncol 2025; 15: 1480247. https://doi.org/10.3389/fonc.2025.1480247
- Karita H, Tsurubuchi T, Amano T, Koiso T, Sakamoto N, Ishikawa E. Spinal cord diffuse midline glioma with postoperative acute swelling: a case report and review of literature. Surg Neurol Int 2023; 14: 360. https://doi.org/10.25259/SNI_636_2023
- Zhang M, Iyer RR, Azad TD, et al. Genomic landscape of intramedullary spinal cord gliomas. Sci Rep 2019; 9: 18722. https://doi.org/10.1038/s41598-019-54286-9
- Schulte JD, Buerki RA, Lapointe S, et al. Clinical, radiologic, and genetic characteristics of histone H3 K27M-mutant diffuse midline gliomas in adults. Neurooncol Adv 2020; 2: vdaa142. https://doi.org/10.1093/noajnl/vdaa142
- Wang YZ, Zhang YW, Liu WH, et al. Spinal cord diffuse midline gliomas with H3 K27m-mutant: clinicopathological features and prognosis. Neurosurgery 2021; 89: 300-307. https://doi.org/10.1093/neuros/nyab174
Copyright and license
Copyright © 2025 The Author(s). This is an open access article distributed under the Creative Commons Attribution License (CC BY), which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is properly cited.






