Öz
Giriş: Servikal disk hernileri, radikülopati ve/veya miyelopatiye yol açabilen yaygın dejeneratif omurga hastalıklarıdır. Bu tabloda en sık tercih edilen cerrahi yöntemlerden biri anterior servikal diskektomi ve füzyon (ASDF)’dur. Ancak ASDF sonrası gelişen komplikasyonlar cerrahi başarıyı ve hasta prognozunu etkileyebilir. Literatürde bildirilen komplikasyon oranları değişkenlik göstermekte, özellikle geniş vaka serili, tek merkezli çalışmaların sayısı sınırlı kalmaktadır. Bu çalışmada, ASDF sonrası komplikasyonlar geniş bir hasta grubunda retrospektif olarak değerlendirilmiştir.
Gereç ve Yöntem: 2012–2023 yılları arasında kliniğimizde ASDF uygulanan, ≥18 yaşındaki ve eksiksiz hasta kayıtlarına sahip 288 hasta retrospektif olarak incelendi. Hastaların yaş, cinsiyet, cerrahi seviye, komplikasyon gelişimi ve komplikasyon türleri kaydedildi. Komplikasyonlar yedi ana kategoride sınıflandırıldı.
Bulgular: Hastaların yaş ortalaması 46,8 yıl olup, kadın-erkek oranı eşittir (1:1). En sık opere edilen düzeyler C5–6 (%33,6) ve C6–7 (%28,1) olarak belirlenmiş, çok seviyeli cerrahiler toplam olguların %29,1’ini oluşturmuştur. Genel komplikasyon oranı %7,63 olarak saptanmıştır. En sık karşılaşılan komplikasyon, reoperasyon gerektiren rezidü disk materyali olup %3,1 oranında gözlenmiştir; bunu %1,7 ile rekürren laringeal sinir hasarı izlemiştir. Postoperatif hematom, nörolojik defisit ve enfeksiyöz komplikasyonlar (apse/diskit) her biri %0,69 oranında tespit edilmiştir. Nadir görülen komplikasyonlar arasında beyin omurilik sıvısı (BOS) fistülü (%0,34) ve inferior tiroid arter yaralanması (%0,34) yer almıştır. Çalışma süresi boyunca özofagus yaralanması veya Horner sendromu olgusu saptanmamıştır. Düşük komplikasyon oranları, cerrahi ilkelere sıkı şekilde uyulması ve standart intraoperatif protokollerin uygulanmasına bağlanabilir.
Sonuç: Literatürdeki birçok çalışmaya kıyasla daha geniş bir hasta serisini kapsayan bu tek merkezli çalışmada, ASDF’nin düşük komplikasyon oranlarıyla güvenli ve etkin bir cerrahi yöntem olduğu gösterilmiştir. Cerrahi prensiplere uygunluk, uygun hasta seçimi ve yakın postoperatif takip, komplikasyonları en aza indirmede kritik rol oynamaktadır.
Anahtar Kelimeler: anterior servikal diskektomi, füzyon cerrahisi, servikal disk hernisi, cerrahi komplikasyonlar
Giriş
Servikal disk hernileri, servikal radikülopati ve/veya miyelopatiye neden olan yaygın dejeneratif omurga hastalıkları arasında yer almaktadır. Bu durumun cerrahi tedavisinde en sık uygulanan yöntem, ilk olarak Smith ve Robinson tarafından tanımlanan anterior servikal diskektomi ve füzyon (ASDF) tekniğidir. Bu prosedürde, disk mesafesine anterior yaklaşımla girilerek dekompresyon sağlanmakta ve ardından interbody füzyon uygulanarak segmental stabilite korunmaktadır.1,2
ASDF, cerrahi etkinliği, yüksek klinik başarı oranları, düşük komplikasyon riski ve minimal postoperatif instabilite ile ilişkili olması nedeniyle servikal disk hastalıklarının tedavisinde altın standart yöntem olarak kabul edilmektedir.3 Literatürde, ASDF’ye bağlı komplikasyon oranlarının %2 ile %15 arasında değiştiği bildirilmektedir. En sık görülen komplikasyonlar arasında rekürren laringeal sinir (RLN) hasarı, disfaji, postoperatif hematom, enfeksiyon, nörolojik defisit, beyin omurilik sıvısı (BOS) fistülü ve vasküler yaralanmalar yer almaktadır.3,4
Komplikasyonların görülme sıklığı; cerrahın deneyimi, hastanın anatomik varyasyonları, uygulanan cerrahi seviye sayısı ve kullanılan implant materyallerine göre farklılık gösterebilir. Özellikle çok seviyeli girişimlerde komplikasyon riskinin arttığı bildirilmektedir.5 Bu çalışmada, kliniğimizde 2012–2023 yılları arasında ASDF uygulanan 288 hastanın verileri retrospektif olarak incelenmiş; operasyon sonrası gelişen komplikasyon türleri ve insidans oranları değerlendirilmiştir. Elde edilen verilerin literatür ile karşılaştırmalı olarak analiz edilmesiyle, klinik pratiğe yön verecek güncel bulguların sunulması amaçlanmaktadır.
Gereç ve Yöntem
Bu retrospektif kesitsel çalışma, 2012–2023 yılları arasında kliniğimizde anterior servikal diskektomi ve füzyon (ASDF) ameliyatı uygulanan, 18 yaş ve üzerindeki, eksiksiz hasta kayıtlarına sahip bireyler arasından seçilmiştir. Her bir hasta için yaş, cinsiyet, cerrahi girişim seviyesi (tek ya da çok seviyeli), etkilenen disk düzeyi, komplikasyon gelişip gelişmediği ve komplikasyonun türü detaylı olarak kaydedilmiştir. Ameliyat sonrası erken dönemde, cage yerleşimi ve füzyon materyallerinin değerlendirilmesi amacıyla tüm hastalara rutin servikal BT görüntülemesi yapılmıştır. BT’de anormal bulgular (örneğin hematom şüphesi) saptanan olgularda ek olarak servikal MRG çekilerek ileri değerlendirme yapılmıştır.
Komplikasyonlar; rekürren laringeal sinir hasarı, postoperatif hematom, nörolojik defisit, apse veya diskit, beyin omurilik sıvısı (BOS) fistülü, inferior tiroid arter düzeyinde vasküler yaralanma ve reoperasyon gerektiren rezidü şeklinde yedi başlık altında sınıflandırılmıştır.
Bulgular
Bu retrospektif çalışmada, 2012–2023 yılları arasında anterior servikal diskektomi ve füzyon (ASDF) uygulanan toplam 288 hastanın verileri incelenmiştir. Hastaların yaş ortalaması 46,8 ± 10,3 yıl olarak belirlenmiş olup, cinsiyet dağılımı kadın ve erkek açısından eşit şekilde (144/144; 1:1 oranında) saptanmıştır (Tablo 1).
| ASDF uygulanan 288 hastanın yaş ortalaması 46,8 ± 10,3 yıl olup, cinsiyet dağılımı kadın ve erkekler arasında eşittir (1:1). Cerrahi müdahale en sık C5–6 (%33,6) ve C6–7 (%28,1) düzeylerinde gerçekleştirilmiştir. Çok seviyeli cerrahi oranı %29,1 olarak belirlenmiştir. | |
| Tablo 1. Hastaların demografik ve cerrahi özellikleri | |
| Özellik | Değer (n=288) |
| Yaş (ortalama ± SD) | 46,8 ± 10,3 yıl |
| Cinsiyet (Kadın/Erkek) | 144 / 144 (1:1) |
| Operasyon seviyesi | |
| C5–6 | 97 (%33,6) |
| C6–7 | 81 (%28,1) |
| C5–6 + C6–7 | 44 (%15,2) |
| C4–5 + C5–6 | 26 (%9) |
| Çok seviyeli cerrahi | 84 (%29,1) |
Cerrahi girişim düzeyleri değerlendirildiğinde, en sık opere edilen segmentin C5–6 (%33,6) olduğu, bunu sırasıyla C6–7 (%28,1) ve C5–6 + C6–7 (%15,2) seviyelerinin izlediği görülmüştür. Daha az sıklıkla C4–5 + C5–6 (%9) seviyelerine müdahale edilmiştir. Çalışma grubunun %29,1’ine çok seviyeli cerrahi uygulanmıştır.
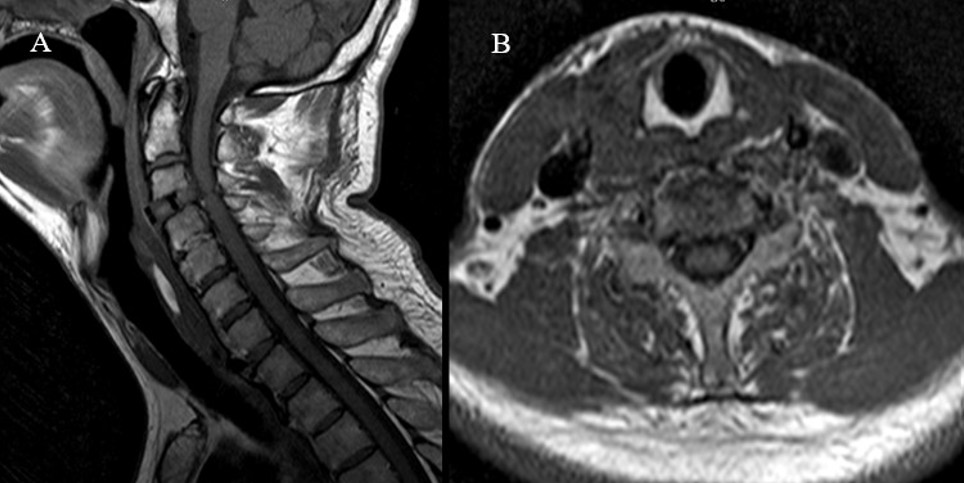
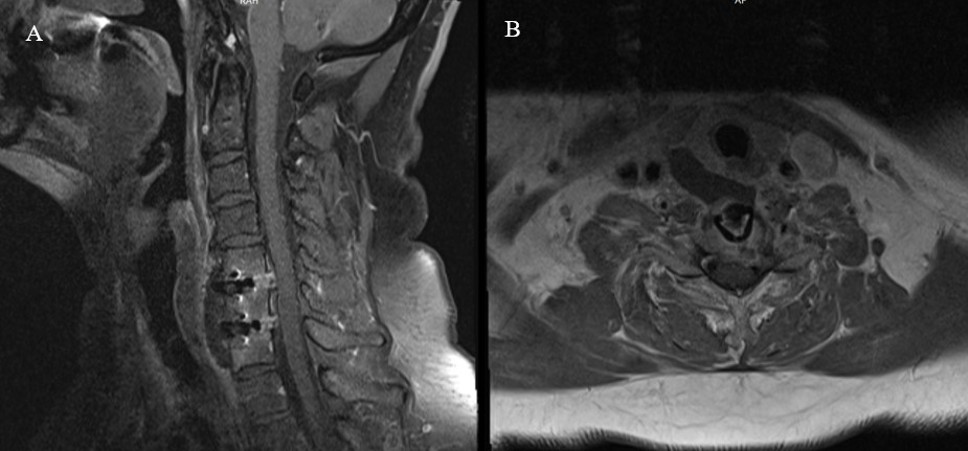
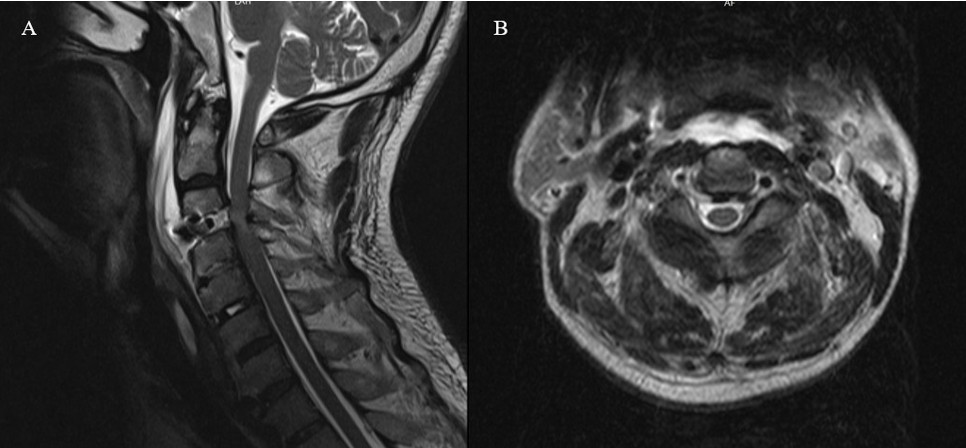
Komplikasyon analizi sonucunda, toplam 22 hastada (%7,63) cerrahiye bağlı komplikasyon geliştiği belirlenmiştir (Tablo 2). En sık karşılaşılan komplikasyon, reoperasyon gerektiren rezidü vakaları olup 9 hastada (%3,1) meydana gelmiştir. Bunu 5 hastada (%1,7) gözlenen rekürren laringeal sinir hasarı takip etmiştir. Postoperatif hematom (Şekil 1), nörolojik defisit ve apse/diskit (Şekil 2) gibi komplikasyonlar her biri %0,69 oranında rapor edilmiştir. Nadir görülen komplikasyonlar arasında BOS fistülü (%0,34) (Şekil 3) ve inferior tiroid arter yaralanması (%0,34) yer almıştır.
| ASDF sonrası gelişen komplikasyon türleri ve oranları sunulmuştur. Toplam komplikasyon oranı %7,63 olarak bulunmuştur. En sık görülen komplikasyon reoperasyon gerektiren rezidü (%3,1), ardından rekürren laringeal sinir hasarı (%1,7) gelmektedir. Diğer komplikasyonlar ise %1’in altında seyretmiş, nadiren BOS fistülü ve vasküler yaralanmalar gözlenmiştir. | ||
| Tablo 2. Komplikasyon türleri ve sıklıkları | ||
| Komplikasyon Türü |
|
|
| Reoperasyon (rezidü nedeniyle) |
|
|
| Rekürren laringeal sinir hasarı |
|
|
| Postoperatif hematom |
|
|
| Nörolojik defisit |
|
|
| Apse / Diskit |
|
|
| BOS fistülü |
|
|
| İnferior tiroid arter yaralanması |
|
|
| Toplam komplikasyon |
|
|
Bu bulgular, ASDF işleminin genel olarak düşük komplikasyon oranlarına sahip olduğunu, ancak bazı risklerin özellikle rezidüel disk materyali ve sinir hasarına bağlı olarak öne çıktığını göstermektedir. Ameliyat sonrası çekilen servikal BT görüntülemelerinde hiçbir hastada füzyon materyallerinin yerinde repozisyon gereksinimi saptanmamıştır. Bu durum, intraoperatif olarak kullanılan skopi rehberliğinin cerrahi doğruluğa katkısı ile açıklanabilir.
Komplikasyon Yönetimi
Reoperasyon Gerektiren Rezidü Disk:
Rezidü disk nedeniyle reoperasyon uygulanan 9 hastada, yeni başlayan veya devam eden radiküler şikayetler üzerine postoperatif ortalama 30. gün (25–40 gün aralığında) içinde kontrol servikal MRG çekilmiştir. Rezidü disk materyali tespit edilen hastalarda, tanı konulmasını takiben ortalama 14 gün (10–20 gün aralığında) içerisinde reoperatif cerrahi müdahale planlanmış ve gerçekleştirilmiştir.
Rekürren Laringeal Sinir Hasarı:
Rekürren laringeal sinir hasarı gelişen 5 hasta Kulak-Burun-Boğaz (KBB) uzmanları tarafından değerlendirildi. Üç hastada konservatif izlem ile 6 ay içinde tam iyileşme sağlandı; iki hastada ise hafif düzeyde kalıcı ses kısıklığı devam etti ve cerrahi müdahale gerektirmedi. Tüm cerrahi yaklaşımlar tek taraflı ve sağdan gerçekleştirilmiş olup, bilateral sinir hasarı gelişmedi. Bu nedenle dispne gelişimi veya trakeostomi gereksinimi olan vaka bulunmadı.
Bizim hasta grubumuzda, postoperatif erken dönemde 1 mg/kg dozunda prednizolon başlanmış ve takip eden haftalarda kademeli doz azaltımı uygulanmıştır. Steroid tedavisi sonrası hastaların büyük çoğunluğunda belirgin iyileşme sağlanmıştır. İyileşmenin temel nedeni, vakalarımızda sinirin tam kat kesilmesi (transeksiyon) değil, cerrahi retraksiyona bağlı geçici kompresyon ve ödem gelişmesidir.
Postoperatif Hematom:
İki hastada (%0,69) servikal cerrahi lojda minimal hematom saptandı. Bu hastalarda postoperatif rutin BT taramalarında hematom şüphesi görülmüş, ileri değerlendirme için kontrastsız servikal MRG çekilerek hematom varlığı doğrulanmıştır. Her iki hastada da solunum sıkıntısı veya nörolojik bulgu gelişmemiştir. Klinik olarak stabil seyreden hastalar konservatif yöntemlerle izlenmiş ve cerrahi drenaja ihtiyaç duyulmamıştır. Hematomlar zamanla kendiliğinden gerilemiştir.
Nörolojik Defisit:
İki hastada postoperatif dönemde motor defisit gelişmiştir. Bir hastada erken fizik tedavi ve rehabilitasyon programı başlanmış ve yaklaşık 3 ay içerisinde tam iyileşme sağlanmıştır. Diğer hastada ise fizik tedaviye rağmen hafif derecede kalıcı güçsüzlük devam etmiştir. Geçici nörolojik defisit saptanan başka olguya rastlanmamıştır.
Apse/Diskit:
İki hastada (%0,69) postoperatif apse ve diskit gelişmiştir. Her iki olguda da klinik enfeksiyon bulguları üzerine yapılan radyolojik değerlendirmede cerrahi lojda koleksiyon ve disk alanında enfeksiyon tespit edilmiştir. Bu hastalarda reoperasyon uygulanmış, füzyon materyalleri çıkarılmış, cerrahi loja geniş irrigasyon yapılmış ve drenaj sağlanmıştır. Operasyon lojuna dren bırakılmıştır. Operasyon sonrası hastalara 8–10 hafta süreyle kültür ve duyarlılık sonuçlarına göre hedefe yönelik intravenöz antibiyotik tedavisi verilmiştir.
Beyin Omurilik Sıvısı (BOS) Fistülü:
Bir hastada (%0,34) postoperatif ilk hafta içerisinde gelişen ortostatik baş ağrısı şikayeti üzerine yapılan görüntülemede BOS fistülü tespit edilmiştir. Bu olguda reoperasyon planlanmış; füzyon materyali çıkarıldıktan sonra dural yırtık alanına dura grefti yerleştirilmiş ve fibrin yapıştırıcı uygulaması yapılmıştır. Ardından tekrar stabilizasyon amacıyla yeni füzyon materyali yerleştirilerek cerrahi alan kapatılmıştır. Hastanın takibinde ek bir komplikasyon gelişmemiştir.
İnferior Tiroid Arter Yaralanması:
Bir hastada (%0,34) cerrahi sırasında tek taraflı inferior tiroid arter yaralanması meydana gelmiştir. Kanama intraoperatif olarak bipolar koterizasyon ve ligasyon yöntemiyle başarıyla kontrol altına alınmıştır. Bizim vakamızda da postoperatif dönemde ek vasküler komplikasyon veya tiroid fonksiyon bozukluğu izlenmemiştir.
Tartışma
Bu çalışmada anterior servikal diskektomi ve füzyon (ASDF) cerrahisi sonrası komplikasyon oranı %7,63 olarak bulunmuştur. Bu oran, literatürde bildirilen %2 ile %15 arasında değişen komplikasyon oranlarıyla uyumlu bir aralıkta yer almaktadır.3,5 Bulgularımız, ASDF’nin genel olarak güvenli bir cerrahi yöntem olduğunu, ancak belirli komplikasyonların dikkate alınması gerektiğini göstermektedir.
Rezidü nedeniyle reoperasyon gereksinimi çalışmamızda %3,1 oranında tespit edilmiştir. Bu oran, literatürde %2 ile %5 arasında değişmekte olup, Fountas ve arkadaşlarının 2007 yılında yayımladığı çalışmada %4 olarak rapor edilmiştir.2 Bulgularımız, söz konusu orana oldukça yakındır ve reoperasyonun ASDF sonrası en sık karşılaşılan komplikasyonlardan biri olduğunu ortaya koymaktadır. Çalışmamızda rezidü disk tanısı ortalama 30. postoperatif günde konulmuş ve tanıyı takiben ortalama 14 gün içerisinde reoperatif cerrahi uygulanmıştır. Erken dönemde yapılan klinik ve radyolojik değerlendirme sayesinde, rezidüel patolojilerin ilerlemeden müdahale edilmesi sağlanmıştır. Bu yaklaşım, nörolojik iyileşmenin hızlanmasına ve cerrahi sonuçların optimize edilmesine katkı sunmuş olabilir. Literatürde de, erken tanı ve müdahale edilen rezidü disk olgularında klinik sonuçların daha başarılı olduğu bildirilmektedir.6-8
Rekürren laringeal sinir hasarı, çalışmamızda %1,7 oranında gözlenmiştir. Literatürde bu komplikasyonun sıklığı %0,2 ile %11 arasında değişmektedir.9,10 Bir sistematik derleme ve meta-analizde bu oranı %3,6 olarak bildirmişti.11 Sonuçlarımız, söz konusu komplikasyonun sıklığının geniş bir aralıkta değişebildiğini ve cerrahi yaklaşım ile hasta anatomisine bağlı olarak farklılaşabileceğini göstermektedir. Postoperatif hematom, çalışmamızda %0,69 oranında görülmüş olup, bu oran literatürde genellikle %0,5 ile %2 arasında bildirilmektedir.12,13 Örneğin, Stienen ve arkadaşları 2015 yılında bu oranı %1,3 olarak raporlamıştır.14
Anatomik olarak değerlendirildiğinde, sağ rekürren laringeal sinir değişken seyir özellikleri gösterirken, sol rekürren laringeal sinir daha doğrusal bir yol izlemektedir. Sağ tarafta bu varyasyonun bulunması, sinir hasarına yatkınlık açısından potansiyel bir risk faktörü oluşturabilir.15
Literatürde rekürren laringeal sinir hasarına yönelik ses terapisi, vokal egzersizler, medializasyon, enjeksiyon laringoplasti ve aritenoid adduksiyon gibi çeşitli tedavi yöntemleri önerilmektedir. Kalıcı bilateral rekürren laringeal sinir hasarı gelişen hastalarda şiddetli dispne durumunda trakeostomi endikasyonu doğabilmektedir. Ancak çalışmamızda, cerrahi yaklaşım ve erken steroid tedavisi sayesinde hiçbir hastada trakeostomi ihtiyacı doğmamış ve iyileşme oranı yüksek olmuştur.16-18
Yeni gelişen nörolojik defisit komplikasyonu ise %0,69 oranında saptanmıştır. Literatürde bu oran %0,3 ile %2 arasında değişmektedir.19,20 Schroeder ve arkadaşlarının 2016 tarihli çalışmasında ise %0,5 olarak bildirilmiştir.21 Bulgularımız, nörolojik defisitlerin cerrahi başarı ile ilişkili olduğunu ve çoğunlukla geçici nitelikte olabileceğini desteklemektedir. Apse ve diskit, çalışmamızda %0,69 oranında gözlenmiştir. Bu komplikasyonun literatürdeki sıklığı genellikle %0,1 ile %1 arasında değişmektedir.13,22 Çalışmamızda gözlenen bu oran, enfeksiyon riskinin düşük olduğunu ancak özellikle sterilizasyon ve cerrahi teknik açısından dikkatli olunması gerektiğini göstermektedir. Beyin omurilik sıvısı (BOS) fistülü komplikasyonu ise yalnızca bir hastada (%0,34) ortaya çıkmıştır. Bu durum literatürde oldukça nadir olarak %0 ile %0,2 arasında bildirilmektedir.23-25 Gözlenen oran, nadir ancak ciddi takip gerektiren bir komplikasyona işaret etmektedir.
İnferior tiroid arter yaralanması da yine %0,34 oranında saptanmıştır ve bu komplikasyon literatürde genellikle %0 ile %0,5 arasında rapor edilmektedir.25-27 Cerrahi sırasında vasküler yapıların korunması bu açıdan kritik önem taşımaktadır. Tek taraflı inferior tiroid arter yaralanmalarında, etkin intraoperatif hemostaz sağlandığında ciddi vasküler komplikasyonlar veya tiroid fonksiyon bozukluğu gelişme riski oldukça düşüktür.28,29 Bizim vakamızda da arter yaralanması cerrahi olarak başarılı şekilde kontrol edilmiş olup, postoperatif dönemde ek vasküler komplikasyon veya tiroid disfonksiyonu izlenmemiştir.
Çalışmamızda özofagus yaralanması görülmemiştir. Ancak bu komplikasyon, ASDF’nin en ciddi ve potansiyel olarak hayati tehlike oluşturan komplikasyonlarından biridir. Geniş serilerde insidansı %0,1 ile %0,3 arasında bildirilmektedir; bazı sistematik derlemelerde bu oran ortalama %0,2 olarak saptanmış, olgu serilerinde %0 ile %1,5 arasında değişen sonuçlara rastlanmıştır. Horner sendromu da çalışmamızda gözlenmemiştir. Bu komplikasyon, iyi huylu ancak nadir görülen bir durumdur. Geniş vaka serilerinde sıklığı %1’in altında raporlanmış, çok merkezli çalışmalarda ise %0,1’in altında dahi görüldüğü bildirilmiştir.30-32
Çalışmamızın bazı kısıtlılıkları bulunmaktadır. Öncelikle, çalışmanın retrospektif tasarımı veri toplamada bazı eksik risk faktörlerinin gözden kaçmasına neden olabilir. İkinci olarak, tek merkezli bir çalışmaya dayanması, elde edilen sonuçların genellenebilirliğini sınırlandırabilir. Ayrıca komplikasyonların değerlendirilmesi, yalnızca kayıtlardaki bilgiler ışığında yapıldığı için hafif düzeydeki geçici sorunlar göz ardı edilmiş olabilir. Prospektif, çok merkezli ve uzun dönem takip verilerini içeren çalışmalar bu konuda daha kapsamlı bilgi sağlayacaktır.
Sonuç
Bu çalışma, anterior servikal diskektomi ve füzyon (ASDF) cerrahisine ait komplikasyon profilini, geniş bir vaka serisi üzerinden, tek merkezli ve cerrahi standartlara uygun uygulamalar temelinde değerlendirmektedir. Literatürdeki birçok çalışmaya kıyasla daha yüksek hasta sayısı ve homojen cerrahi yaklaşımı sayesinde, elde edilen veriler ASDF’nin güvenliğini ve uygulanabilirliğini güçlü biçimde desteklemektedir. Komplikasyon oranlarının düşük seviyelerde seyretmesi, cerrahi tekniklerin doğru uygulanmasının ve hasta takibinin önemini bir kez daha ortaya koymaktadır. Bu sonuçlar, klinik pratiğe katkı sağlamayı ve ASDF’nin komplikasyon profiline dair daha net bir çerçeve sunmayı amaçlamaktadır. Çalışmamız, bu alanda yapılan yüksek nitelikli vaka serilerine dayalı araştırmaların önemini vurgulamakta ve ASDF’nin cerrahi başarı açısından güvenilir bir yöntem olduğunu göstermektedir.
Etik kurul onayı
Bu çalışma retrospektif bir tasarıma sahip olduğundan, insan katılımcılardan doğrudan veri toplanmamış ve müdahale gerçekleştirilmemiştir; bu nedenle etik kurul onayı alınmamıştır.
Yazarlık katkısı
Çalışma konsepti ve tasarımı: VÖ, ME, DŞ, PAS, AA, YA, AS; veri toplama: VÖ, ME; sonuçların analizi ve yorumlanması: CİG, DD, İD; makaleyi hazırlama: CİG, DD, PAS, AA, YA, AS. Yazar(lar) sonuçları gözden geçirmiş ve makalenin son halini onaylamıştır.
Finansman
Yazar(lar), çalışmanın herhangi bir finansal destek almadığını beyan etmiştir.
Çıkar çatışması
Yazar(lar) herhangi bir çıkar çatışması olmadığını beyan etmiştir.
Kaynakça
- Cloward RB. The anterior approach for removal of ruptured cervical disks. J Neurosurg 1958; 15: 602-617. https://doi.org/10.3171/jns.1958.15.6.0602
- Fountas KN, Kapsalaki EZ, Nikolakakos LG, et al. Anterior cervical discectomy and fusion associated complications. Spine (Phila Pa 1976) 2007; 32: 2310-2317. https://doi.org/10.1097/BRS.0b013e318154c57e
- Yee TJ, Swong K, Park P. Complications of anterior cervical spine surgery: a systematic review of the literature. J Spine Surg 2020; 6: 302-322. https://doi.org/10.21037/jss.2020.01.14
- Tasiou A, Giannis T, Brotis AG, et al. Anterior cervical spine surgery-associated complications in a retrospective case-control study. J Spine Surg 2017; 3: 444-459. https://doi.org/10.21037/jss.2017.08.03
- Epstein NE. A review of complication rates for Anterior Cervical Diskectomy and Fusion (ACDF). Surg Neurol Int 2019; 10: 100. https://doi.org/10.25259/SNI-191-2019
- Liang W, Xiong Y, Jia Y, et al. Anterior cervical discectomy and fusion for the treatment of giant cervical disc herniation. J Orthop Surg Res 2023; 18: 683. https://doi.org/10.1186/s13018-023-04036-5
- Watkins RG, Watkins RG. Cervical disc herniations, radiculopathy, and myelopathy. Clin Sports Med 2021; 40: 513-539. https://doi.org/10.1016/j.csm.2021.03.006
- Mostofi K, Khouzani RK. Reliability of cervical radiculopathy, its congruence between patient history and medical imaging evidence of disc herniation and its role in surgical decision. Eur J Orthop Surg Traumatol 2016; 26: 805-808. https://doi.org/10.1007/s00590-016-1801-z
- Erwood MS, Hadley MN, Gordon AS, Carroll WR, Agee BS, Walters BC. Recurrent laryngeal nerve injury following reoperative anterior cervical discectomy and fusion: a meta-analysis. J Neurosurg Spine 2016; 25: 198-204. https://doi.org/10.3171/2015.9.SPINE15187
- Debkowska MP, Butterworth JF, Moore JE, Kang S, Appelbaum EN, Zuelzer WA. Acute post-operative airway complications following anterior cervical spine surgery and the role for cricothyrotomy. J Spine Surg 2019; 5: 142-154. https://doi.org/10.21037/jss.2019.03.01
- Yerneni K, Burke JF, Chunduru P, et al. Safety of outpatient anterior cervical discectomy and fusion: a systematic review and meta-analysis. Neurosurgery 2020; 86: 30-45. https://doi.org/10.1093/neuros/nyy636
- Cherry C. Anterior cervical discectomy and fusion for cervical disc disease. AORN J 2002; 76: 996-1008. https://doi.org/10.1016/s0001-2092(06)61001-6
- Quinto ES, Paisner ND, Huish EG, Senegor M. Ten-year outcomes of cervical disc arthroplasty versus anterior cervical discectomy and fusion : a systematic review with meta-analysis. Spine (Phila Pa 1976) 2024; 49: 463-469. https://doi.org/10.1097/BRS.0000000000004887
- Stienen MN, Joswig H, Jucker D, Hildebrandt G, Schaller K, Gautschi OP. Anterior cervical discectomy and fusion: is surgical education safe? Acta Neurochir (Wien) 2015; 157: 1395-1404. https://doi.org/10.1007/s00701-015-2396-6
- Thomas AM, Fahim DK, Gemechu JM. Anatomical variations of the recurrent laryngeal nerve and implications for injury prevention during surgical procedures of the neck. Diagnostics (Basel) 2020; 10: 670. https://doi.org/10.3390/diagnostics10090670
- Oh LJ, Dibas M, Ghozy S, Mobbs R, Phan K, Faulkner H. Recurrent laryngeal nerve injury following single- and multiple-level anterior cervical discectomy and fusion: a meta-analysis. J Spine Surg 2020; 6: 541-548. https://doi.org/10.21037/jss-20-508
- Echevarria AC, Hershfeld B, Verma R, Bruni M. Recurrent laryngeal nerve injury during Anterior Cervical Discectomy and Fusion (ACDF): a case presentation and review of the literature. Cureus 2024; 16: e64603. https://doi.org/10.7759/cureus.64603
- Strohl MP, Choy W, Clark AJ, et al. Immediate voice and swallowing complaints following revision anterior cervical spine surgery. Otolaryngol Head Neck Surg 2020; 163: 778-784. https://doi.org/10.1177/0194599820926133
- Hardman M, Bhandarkar AR, Jarrah RM, Bydon M. Predictors of airway, respiratory, and pulmonary complications following elective anterior cervical discectomy and fusion. Clin Neurol Neurosurg 2022; 217: 107245. https://doi.org/10.1016/j.clineuro.2022.107245
- Wright IP, Eisenstein SM. Anterior cervical discectomy and fusion without instrumentation. Spine (Phila Pa 1976) 2007; 32: 772-774. https://doi.org/10.1097/01.brs.0000258846.86537.ad
- Schroeder GD, Kurd MF, Millhouse PW, Vaccaro AR, Hilibrand AS. Performing an Anterior Cervical Discectomy and Fusion. Clin Spine Surg 2016; 29: 186-190. https://doi.org/10.1097/BSD.0000000000000383
- Thomas G, Gupta P, Chaudhry T, et al. Impact of obesity on Anterior Cervical Discectomy and Fusion (ACDF): postoperative morbidity and mortality. Turk Neurosurg 2023; 33: 1047-1052. https://doi.org/10.5137/1019-5149.JTN.43115-22.1
- McClelland S, Oren JH, Protopsaltis TS, Passias PG. Outpatient anterior cervical discectomy and fusion: a meta-analysis. J Clin Neurosci 2016; 34: 166-168. https://doi.org/10.1016/j.jocn.2016.06.012
- Charalampidis A, Hejrati N, Ramakonar H, Kalsi PS, Massicotte EM, Fehlings MG. Clinical outcomes and revision rates following four-level anterior cervical discectomy and fusion. Sci Rep 2022; 12: 5339. https://doi.org/10.1038/s41598-022-09389-1
- Robertson SC, Ashley MR. Complications of Anterior Cervical Discectomy and Fusion. Acta Neurochir Suppl 2023; 130: 169-178. https://doi.org/10.1007/978-3-030-12887-6_20
- Nachalon Y. Anterior cervical spine surgery and dysphagia. Curr Opin Otolaryngol Head Neck Surg 2022; 30: 417-421. https://doi.org/10.1097/MOO.0000000000000845
- Yang Z, Zhao Y, Luo J. Incidence of dysphagia of zero-profile spacer versus cage-plate after anterior cervical discectomy and fusion: A meta-analysis. Medicine (Baltimore) 2019; 98: e15767. https://doi.org/10.1097/MD.0000000000015767
- Debek AH, Jammaleddine GW, Bou-Khalil PK. Inferior thyroid artery injury after attempts of internal jugular venous catheterization. J Cardiothorac Vasc Anesth 2007; 21: 422-424. https://doi.org/10.1053/j.jvca.2006.05.010
- Petrocheilou G, Myrianthefs P, Evodia E, Vlychou M, Kokkinis CE. Inferior thyroid artery pseudoaneurysm caused by blunt trauma. Vascular 2010; 18: 242-245. https://doi.org/10.2310/6670.2010.00030
- Lubelski D, Pennington Z, Sciubba DM, Theodore N, Bydon A. Horner syndrome after anterior cervical discectomy and fusion: case series and systematic review. World Neurosurg 2020; 133: e68-e75. https://doi.org/10.1016/j.wneu.2019.08.101
- Vavruch L, Hedlund R, Javid D, Leszniewski W, Shalabi A. A prospective randomized comparison between the cloward procedure and a carbon fiber cage in the cervical spine: a clinical and radiologic study. Spine (Phila Pa 1976) 2002; 27: 1694-1701. https://doi.org/10.1097/00007632-200208150-00003
- Riley LH, Vaccaro AR, Dettori JR, Hashimoto R. Postoperative dysphagia in anterior cervical spine surgery. Spine (Phila Pa 1976) 2010; 35: S76-S85. https://doi.org/10.1097/BRS.0b013e3181d81a96
Telif hakkı ve lisans
Telif hakkı © 2025 Yazar(lar). Açık erişimli bu makale, orijinal çalışmaya uygun şekilde atıfta bulunulması koşuluyla, herhangi bir ortamda veya formatta sınırsız kullanım, dağıtım ve çoğaltmaya izin veren Creative Commons Attribution License (CC BY) altında dağıtılmıştır.






